What happens to the hydrogen concentration in the thylakoid and why is this important?
5.2: The Light-Dependent Reactions of Photosynthesis
Learning Objectives
Past the terminate of this section, you will exist able to:
- Explicate how plants absorb energy from sunlight
- Describe how the wavelength of light affects its free energy and color
- Describe how and where photosynthesis takes identify within a plant
How can light be used to make nutrient? It is easy to remember of light as something that exists and allows living organisms, such as humans, to see, just calorie-free is a grade of energy. Similar all energy, light can travel, modify class, and be harnessed to practise work. In the case of photosynthesis, lite energy is transformed into chemical free energy, which autotrophs use to build carbohydrate molecules. Notwithstanding, autotrophs only use a specific component of sunlight (Figure 5.eight).

Concept in Activeness

Visit this site and click through the animation to view the process of photosynthesis within a foliage.
What Is Lite Free energy?
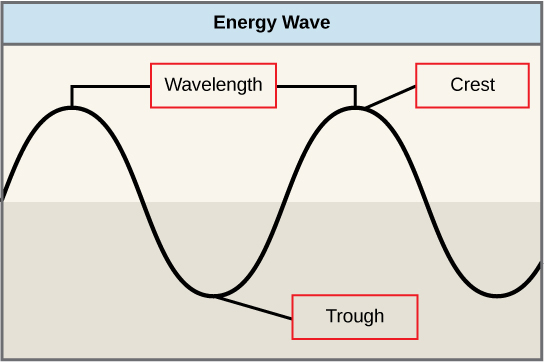
The sun emits an enormous amount of electromagnetic radiations (solar energy). Humans tin can see only a fraction of this energy, which is referred to as "visible light." The style in which solar energy travels can exist described and measured equally waves. Scientists can determine the amount of energy of a moving ridge by measuring its wavelength, the distance between 2 sequent, similar points in a series of waves, such as from crest to crest or trough to trough (Figure v.ix).
Visible low-cal constitutes merely one of many types of electromagnetic radiation emitted from the sun. The electromagnetic spectrum is the range of all possible wavelengths of radiation (Figure 5.10). Each wavelength corresponds to a dissimilar amount of energy carried.
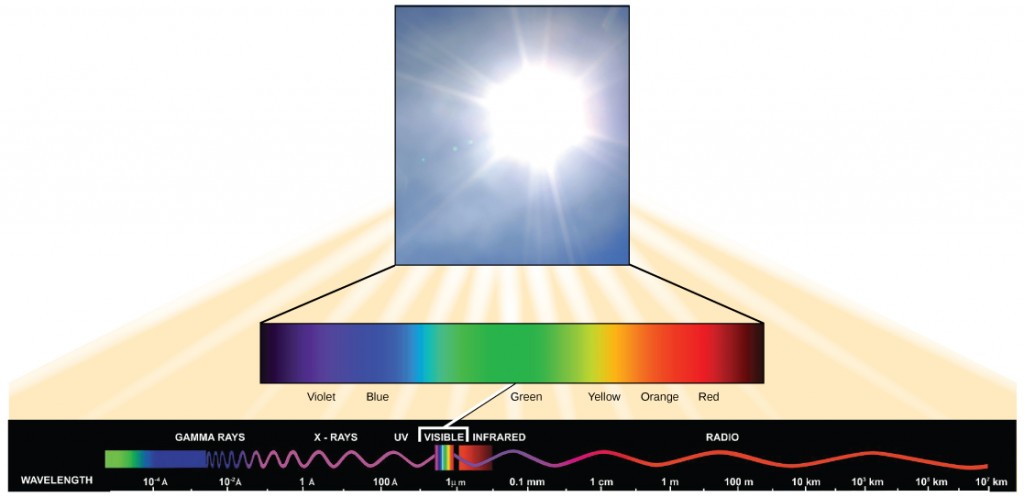
Each type of electromagnetic radiation has a feature range of wavelengths. The longer the wavelength (or the more stretched out it appears), the less energy is carried. Short, tight waves deport the most energy. This may seem illogical, simply think of it in terms of a piece of moving rope. It takes little effort past a person to motion a rope in long, wide waves. To make a rope move in brusque, tight waves, a person would need to apply significantly more energy.
The dominicus emits a broad range of electromagnetic radiation, including X-rays and ultraviolet (UV) rays. The higher-energy waves are dangerous to living things; for case, X-rays and UV rays can exist harmful to humans.
Absorption of Light
Lite energy enters the process of photosynthesis when pigments absorb the light. In plants, pigment molecules blot simply visible calorie-free for photosynthesis. The visible lite seen by humans as white light actually exists in a rainbow of colors. Certain objects, such every bit a prism or a drib of water, disperse white light to reveal these colors to the man eye. The visible light portion of the electromagnetic spectrum is perceived by the human eye as a rainbow of colors, with violet and blue having shorter wavelengths and, therefore, higher free energy. At the other end of the spectrum toward ruby-red, the wavelengths are longer and have lower energy.
Understanding Pigments
Dissimilar kinds of pigments exist, and each absorbs only certain wavelengths (colors) of visible low-cal. Pigments reflect the colour of the wavelengths that they cannot blot.
All photosynthetic organisms contain a paint called chlorophyll a, which humans see as the common green color associated with plants. Chlorophyll a absorbs wavelengths from either end of the visible spectrum (blueish and red), but not from green. Because green is reflected, chlorophyll appears green.
Other pigment types include chlorophyll b (which absorbs blue and crimson-orangish lite) and the carotenoids. Each type of pigment can be identified past the specific pattern of wavelengths it absorbs from visible low-cal, which is its absorption spectrum.
Many photosynthetic organisms take a mixture of pigments; between them, the organism can blot energy from a wider range of visible-calorie-free wavelengths. Non all photosynthetic organisms have full access to sunlight. Some organisms grow underwater where light intensity decreases with depth, and certain wavelengths are captivated by the water. Other organisms grow in competition for light. Plants on the rainforest floor must exist able to absorb any flake of light that comes through, because the taller copse cake near of the sunlight (Figure five.11).

How Light-Dependent Reactions Work
The overall purpose of the light-dependent reactions is to convert light energy into chemic energy. This chemic energy will be used past the Calvin cycle to fuel the associates of saccharide molecules.
The low-cal-dependent reactions begin in a group of pigment molecules and proteins called a photosystem. Photosystems be in the membranes of thylakoids. A pigment molecule in the photosystem absorbs i photon, a quantity or "packet" of light energy, at a time.
A photon of light energy travels until information technology reaches a molecule of chlorophyll. The photon causes an electron in the chlorophyll to go "excited." The free energy given to the electron allows it to break free from an atom of the chlorophyll molecule. Chlorophyll is therefore said to "donate" an electron (Figure 5.12).
To replace the electron in the chlorophyll, a molecule of water is split. This splitting releases an electron and results in the formation of oxygen (Otwo) and hydrogen ions (H+) in the thylakoid space. Technically, each breaking of a water molecule releases a pair of electrons, and therefore tin replace two donated electrons.
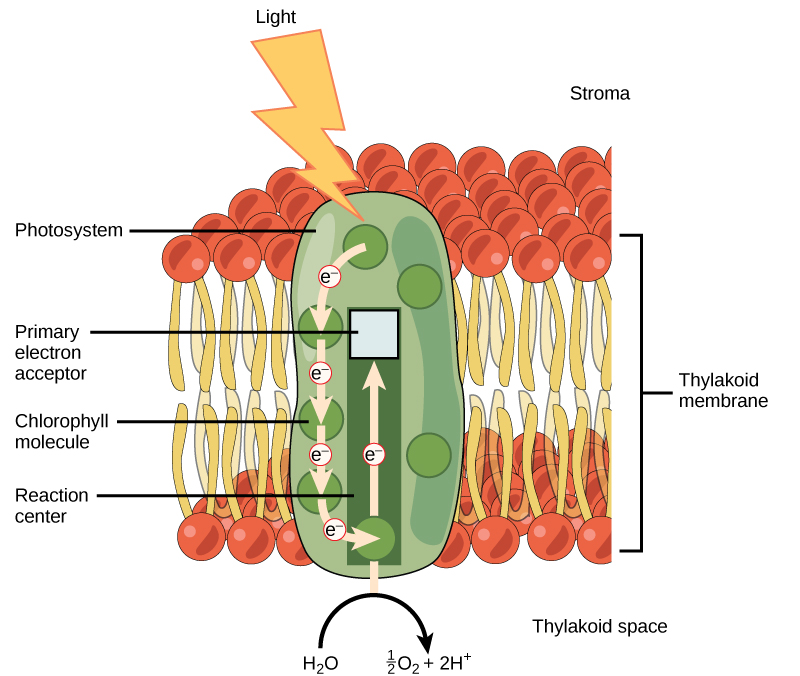
The replacing of the electron enables chlorophyll to respond to some other photon. The oxygen molecules produced equally byproducts find their way to the surrounding environment. The hydrogen ions play critical roles in the balance of the low-cal-dependent reactions.
Keep in mind that the purpose of the light-dependent reactions is to convert solar free energy into chemical carriers that volition be used in the Calvin bicycle. In eukaryotes and some prokaryotes, two photosystems be. The starting time is chosen photosystem II, which was named for the order of its discovery rather than for the order of the function.
After the photon hits, photosystem II transfers the free electron to the first in a series of proteins inside the thylakoid membrane called the electron transport chain. As the electron passes along these proteins, energy from the electron fuels membrane pumps that actively move hydrogen ions confronting their concentration gradient from the stroma into the thylakoid space. This is quite analogous to the procedure that occurs in the mitochondrion in which an electron transport chain pumps hydrogen ions from the mitochondrial stroma across the inner membrane and into the intermembrane space, creating an electrochemical gradient. Later on the energy is used, the electron is accepted by a pigment molecule in the next photosystem, which is called photosystem I (Figure 5.13).
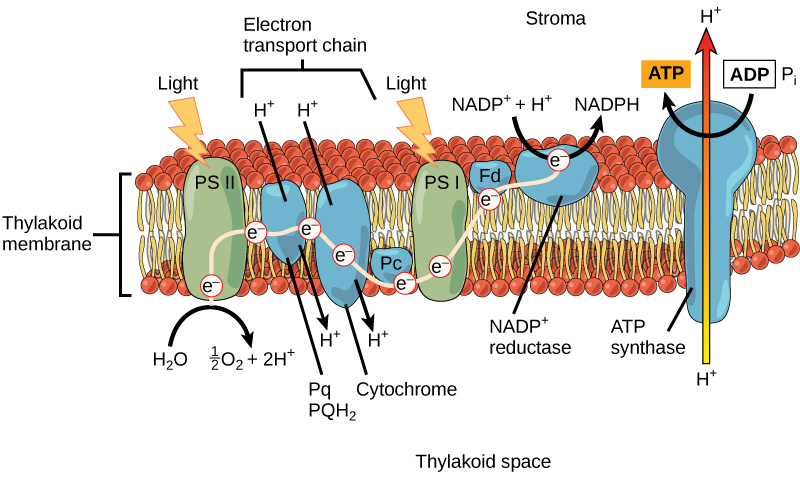
Generating an Energy Carrier: ATP
In the light-dependent reactions, energy absorbed by sunlight is stored by two types of energy-carrier molecules: ATP and NADPH. The energy that these molecules carry is stored in a bond that holds a unmarried cantlet to the molecule. For ATP, it is a phosphate atom, and for NADPH, it is a hydrogen atom. Recall that NADH was a like molecule that carried energy in the mitochondrion from the citric acid bike to the electron transport concatenation. When these molecules release energy into the Calvin bike, they each lose atoms to become the lower-energy molecules ADP and NADP+.
The buildup of hydrogen ions in the thylakoid space forms an electrochemical gradient because of the difference in the concentration of protons (H+) and the difference in the charge across the membrane that they create. This potential energy is harvested and stored as chemic energy in ATP through chemiosmosis, the motility of hydrogen ions down their electrochemical slope through the transmembrane enzyme ATP synthase, just as in the mitochondrion.
The hydrogen ions are immune to laissez passer through the thylakoid membrane through an embedded poly peptide complex chosen ATP synthase. This same protein generated ATP from ADP in the mitochondrion. The free energy generated past the hydrogen ion stream allows ATP synthase to attach a third phosphate to ADP, which forms a molecule of ATP in a process called photophosphorylation. The period of hydrogen ions through ATP synthase is chosen chemiosmosis, because the ions motion from an area of high to low concentration through a semi-permeable construction.
Generating Another Energy Carrier: NADPH
The remaining function of the light-dependent reaction is to generate the other energy-carrier molecule, NADPH. As the electron from the electron transport concatenation arrives at photosystem I, it is re-energized with some other photon captured by chlorophyll. The energy from this electron drives the formation of NADPH from NADP+ and a hydrogen ion (H+). Now that the solar energy is stored in energy carriers, it tin exist used to make a sugar molecule.
Section Summary
In the beginning office of photosynthesis, the light-dependent reaction, pigment molecules absorb free energy from sunlight. The nigh common and arable pigment is chlorophyll a. A photon strikes photosystem Two to initiate photosynthesis. Energy travels through the electron transport chain, which pumps hydrogen ions into the thylakoid infinite. This forms an electrochemical gradient. The ions flow through ATP synthase from the thylakoid infinite into the stroma in a process called chemiosmosis to form molecules of ATP, which are used for the germination of sugar molecules in the 2d stage of photosynthesis. Photosystem I absorbs a second photon, which results in the formation of an NADPH molecule, another free energy carrier for the Calvin cycle reactions.
Glossary
assimilation spectrum: the specific pattern of absorption for a substance that absorbs electromagnetic radiations
chlorophyll a: the form of chlorophyll that absorbs violet-bluish and cerise light
chlorophyll b: the class of chlorophyll that absorbs blue and scarlet-orange light
electromagnetic spectrum: the range of all possible frequencies of radiation
photon: a distinct quantity or "packet" of low-cal energy
photosystem: a group of proteins, chlorophyll, and other pigments that are used in the calorie-free-dependent reactions of photosynthesis to blot lite energy and catechumen information technology into chemical energy
wavelength: the distance between consecutive points of a wave
Source: https://opentextbc.ca/biology/chapter/5-2-the-light-dependent-reactions-of-photosynthesis/
0 Response to "What happens to the hydrogen concentration in the thylakoid and why is this important?"
Postar um comentário